 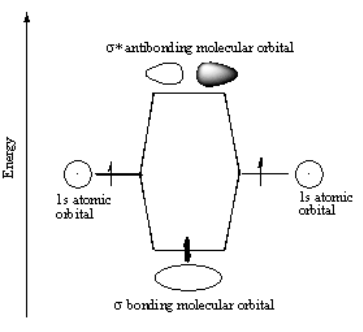
EISA and collision-induced dissociation (CID) among other physical events that impact ions are a part of gas-phase ion chemistry. Tandem mass spectrometry-generated fragmentation is typically made in the collision zone (post-source fragmentation) of a tandem mass spectrometer. Prior to these experiments, electrospray ionization in-source fragmentation was generally considered an undesired effect however, electrospray ionization using Enhanced In-Source Fragmentation/Annotation (EISA) has been shown to promote in-source fragmentation that creates fragment ions that are consistent with tandem mass spectrometers. When chemical bonds are created, species that participate in them are more stable, usually because they have an imbalance of charge (more or fewer electrons than protons) or because their valence electrons do not fill or half-fill electron orbitals.Toluene Fragmentation Mass spectrometry techniques įragmentation can occur in the ion source (in-source fragmentation) where it has been used with electron ionization to help identify molecules and, recently (2020), with electrospray ionization it has been shown to provide the same benefit in facilitating molecular identification. The electrons of an atom or ion are pulled to its positive charge (which contains protons), as well as the nuclei of nearby atoms. The preponderance of bonding activity can be explained by the interaction between two distinct electrostatic forces. Sigma bonds form in all alkanes, alkenes, and alkynes.Typically, the sigma bond is represented by the sign.When two atoms or molecules engage with one another, the initial step is the development of a sigma bond.The sigma bond is extremely strong and long-lasting.Head-on-head overlapping of hybrid orbitals forms the sigma bond.and overlap, for example, sigma bond’s characteristics They’re made up of s-s overlaps, such as, and overlaps, such as. orbitals must be aligned along the internuclear axis to establish a sigma bond. As a result, overlap always results in a sigma bond. Because orbitals are asymmetric, they can overlap on any side. The language and tools of symmetry groups are most precisely defined for diatomic molecules when it comes to sigma bonding.Ī sigma-bond is symmetrical with respect to rotation about the bond axis in this formal method.Ī sigma link is a covalent bond created by collinear or coaxial overlapping of an atomic orbital in a line of internuclear axis. Sigma bonds (bonds) are a form of covalently chemical bond in chemistry. The triple covalent bond can be found in the acetylene molecule. Bonding in oxygen, carbon dioxide, and ethylene molecules, for example, is produced by numerous covalent bonds. When two or three electron pairs are shared by two or three atoms, these types of bonds are produced. Multiple bonds are made up of double and triple covalent bonds. Lewis’s portrayal of numerous covalent molecular bonds was also expanded. Two chlorine atoms combine to produce the covalent chlorine molecule, according to Lewis.Įach atom contributes electrons to form a pair that is shared by both atoms through covalent chemical bonding. In the outermost quantum energy level, a chlorine atom has seven electrons. Just one pair of electrons is shared in between linked atoms to form a single covalent bond. The covalent bond can be one of three forms depending on the number of electron pairs. Van der Waal forces of attraction: Atoms are attracted to one another and form bonds.Hydrogen bond: A hydrogen atom joins several of the most electronegative atoms, including oxygen, nitrogen, and fluorine.Covalent bond: A covalent bond is formed by sharing of electrons.Ionic bond: An ionic link is formed when all of the electrons in a molecule have been exchanged.The ways by which a chemical bonding can take place are mentioned below: A chemical bond is implied by the bound state, which suggests a net attractive force between the atoms. When the overall energy of the combination is smaller than that of the energy of the isolated atoms, the compound is viable. Two or more atoms are joined together to produce chemical compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
